NEW YORK — A recall of the emergency anti-allergy medicine EpiPen is expanding to the U.S. and other markets in North America, Europe, Asia and South America because the allergy shots may not work.
The notice issued Friday by Mylan N.V. expands upon warnings made earlier this month after two reports of the device failing. The recall now covers 13 lots distributed from December 2015 through July 2016.
Mylan didn’t immediately respond to a question about how many devices are affected by the recall.
The problems could be potentially life-threatening, although Mylan described the incidence of the defects as “extremely rare.”
The EpiPen is used to treat allergic reactions to certain food and bug bites
The products can be replaced at no charge by calling 877-650-3494 or emailing customer.service@mylan.com.
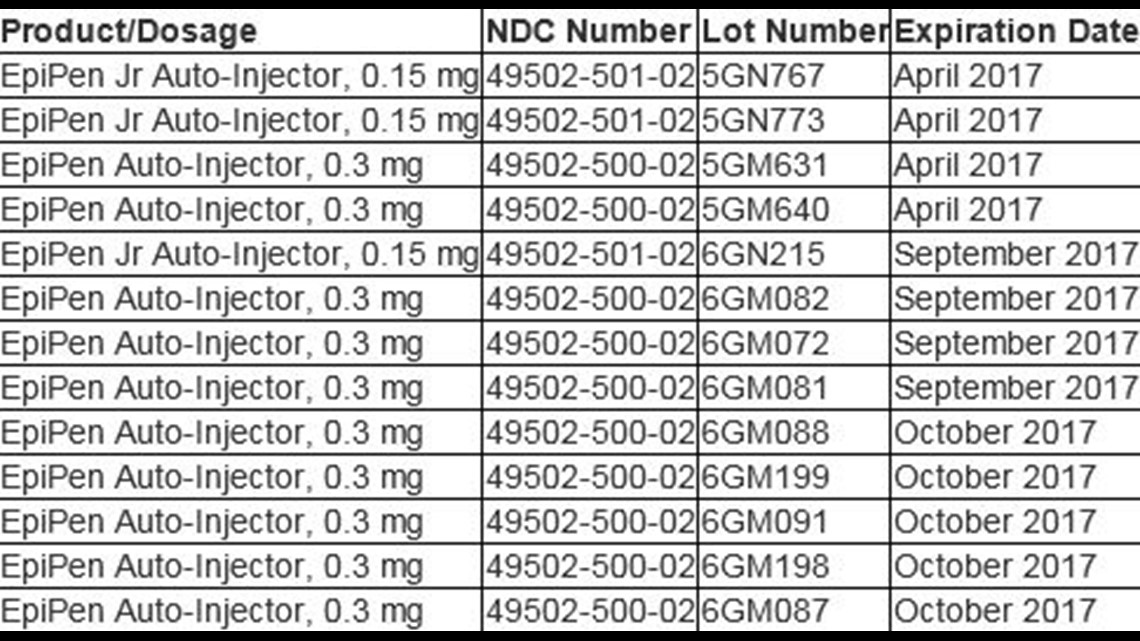
At this time, the 13 lots identified – distributed between Dec. 17, 2015, and July 1, 2016 – are the only EpiPen lots impacted by the U.S. recall. Consumers who have EpiPens from lots that are not included in this recall, do not need to replace their EpiPen prior to its expiration date.
Below is a list of the 13 EpiPen lots impacted by the U.S. recall.